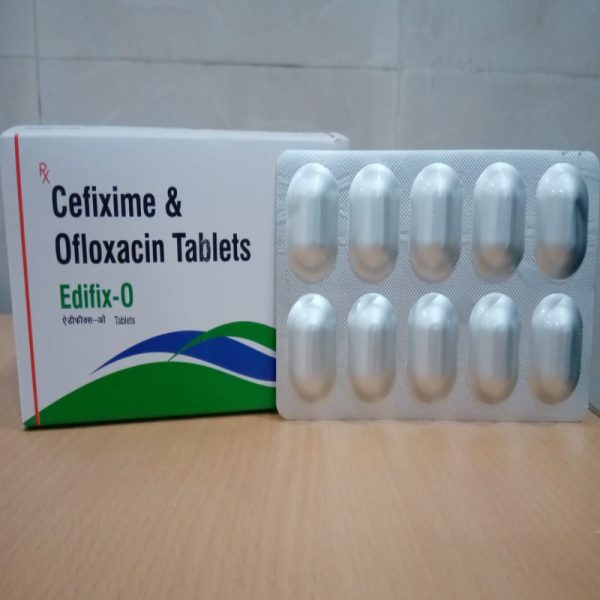
EDIFIX-O TABLET
When Infections are dominating
Composition:
Each film coated tablet contains:
Cefixime 200mg, Ofloxacin 200mg
CEFIXIME:
It is third generation oral cephalosporin,effective against a wide spectrum of sensitive gram positive, gram negative & anaerobic bacterial pathogens including beta-lactamase producing strains.It has high affinity for penicillin-binding proteins with varying site of activity. It acts by inhibition of bacterial cell-wall synthesis. The elimination half-life is about 3 hours, with little variation over the usual therapeutic dosage range.
Indication of CEFIXIME:
Infections of respiratory, urinary & biliary tract.
Contra-Indications
Hypersensitivity.
Special Precautions
Hypersensitivity to penicillins. Severe renal impairment. Paediatrics:Safety upto 6 months age not established. Pregnancy: Use with caution. Lactation: Use with caution.
Side Effects
G-I disturbances. Headaches, dizziness. Rashes.
Drug Interactions
Salicylic acid & Probenecid.
Indications of OFLOXACIN:
Genito urinary, respiratory, Gastro-intestinal, Skin & Soft tissue infections. Peritonitis, Gonorrhoea.
Contra-Indications:
Hypersensitivity to 4-quinolones. History of epilepsy.
Special Precautions:
Exposure to strong sun light and ultra violet rays should be avoided. Driving or operating machinery. Renal insufficiency, Psychiatric disorders concomitant use of aluminium and magnesium containing antacids. Paediatrics: Safety not established in children below 16 years. Pregnancy: Contraindicated. Lactation: Contraindicated. Elderly: Safe.
Side Effects:
Hypersensitivity reactions,skin reactions, CNS disturbances,G.I upset pseudomembranous colitis.Transient increase in liver enzymes. Rarely, joint /muscle pain,bone marrow depression.
Drug Interactions:
Antacids containing magnesium and aluminium reduce absorption. Sucralfate reduces absorption. Inhibits GABA receptor binding. Probenecid decreases elimination.
Packaging:
Pack (10×10 Tab)

Reviews
There are no reviews yet.